Whether researchers are studying pediatric nervous systems in child neurology or looking at potential cancer treatments, eliminating the possibility of bias is essential to get definitive answers to difficult medical questions.
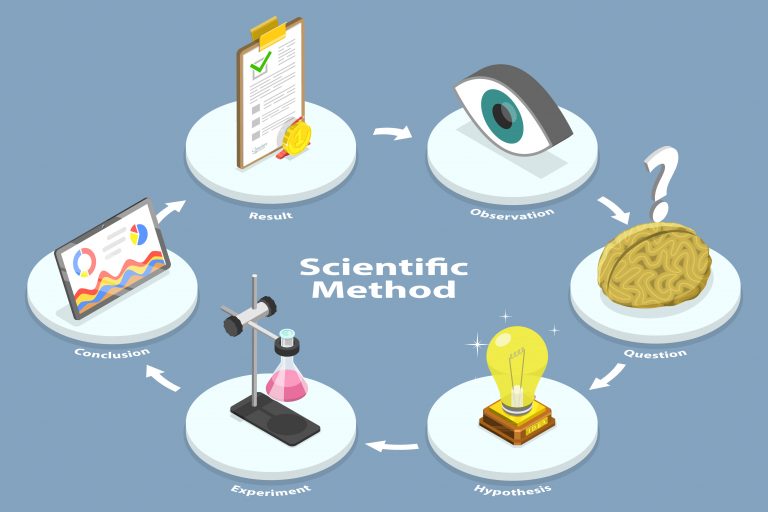
One of the key ways that researchers eliminate bias is through a process called trial randomization. Here, we’ll explore how clinical randomization works to prevent bias, why it’s important, and how randomization improves clinical investigations into exciting new treatments.
What Is Bias in Clinical Trials?
Often, when we hear the word “bias” in everyday contexts, our minds immediately jump to common social challenges like racism or sexism. And, while eliminating possible racial biases, etc. is important for clinical trials, the term is actually much more expansive.
The American Journal of Epidemiology defines bias in clinical trials as:
“Systematic errors that encourage one outcome over others. The potential effect of bias is that investigators will come to the wrong conclusions about the beneficial and harmful effects of interventions.”
As the definition explains, a biased study can come to wrongful conclusions, potentially in many cases with life-or-death implications for physicians who rely on their results to treat patients with serious health conditions.
Preventing bias in clinical trials, therefore, is fundamental to a solid study design. Before we talk about strategies researchers employ to prevent bias from threatening clinical trials, let’s briefly explore the most frequent types of bias that present themselves.
Three Types of Bias in Clinical Trials
The three types of bias in clinical trials are:
- Selection bias. This error occurs in the very first stages of a trial when participants are selected. To accurately answer the clinical issue under study, the human participants must represent the target population that the researchers intend to study. There are various forms of selection bias in clinical research, including the non-response bias, the incidence-prevalence bias, the loss-to-follow-up bias, the confounding by indication bias, and the volunteer bias.
- Classification bias. Sometimes known alternatively as information bias, classification bias can occur at any point in a flawed research design. It most often occurs when clinical participants are assigned to a category they shouldn’t belong to. For example, if a patient with high blood pressure who is receiving treatment and therefore displays normal blood pressure readings is assigned to a “normal blood pressure” group in a study when he or she should rightly be classified in the “high blood pressure” group, that is classification bias.
- Confounding bias. The term “confound” means “to fail to discern differences between: mix up.” In clinical research, confounding bias refers to a “systematic distortion in the measure of association between exposure and the health outcome caused by mixing the effect of the exposure of primary interest with extraneous risk factors.”
What Is Randomization in Clinical Trials?
In its simplest explanation, randomization controls bias-inducing factors in an experiment that would otherwise be uncontrollable given the study design without randomization measures. A clinical trial that employs one or more randomization techniques is called a randomized controlled trial (RCT).
Here’s a diagram of a very simple randomization design:

The US Department of Health and Human Services (HHS) explains randomization in this helpful video below:
The benefit of randomization is the elimination of bias:
“Randomized controlled trial is widely accepted as the best design for evaluating the efficacy of a new treatment because of the advantages of randomization (random allocation). Randomization eliminates accidental bias, including selection bias.”
How Randomization Prevents Bias in Clinical Research
Researchers use randomization to prevent bias in clinical research projects by ensuring that there is no undue influence, conscious or otherwise, either on the part of the researchers or the study participants in allocating them to one group or another (traditionally, control vs. treatment group):
“[Randomization] prevents the selection bias and insures against the accidental bias. It produces the comparable groups and eliminates the source of bias in treatment assignments. Finally, it permits the use of probability theory to express the likelihood of chance as a source for the difference of end outcome.”
Essentially, randomization eliminates human choice from the assignment process – because, as we all know, even the most professional, competent, well-intentioned humans are prone to error. Trial volunteers and even top-tier researchers are not exempt.
The Types of Randomization in Clinical Trials
Clinical researchers achieve randomization through various means, which we’ll explore now. Researchers use one of three main methods to randomize trials, ranging from basic to complex:
- Simple randomization. This, as the name suggests, is the most straightforward, least labor-intensive randomization technique available to researchers. In clinical trials with two groups (control group and the group receiving the real treatment), researchers can assure total randomization by simply flipping a coin, drawing cards, or rolling dice. Simple randomization works best for large-scale studies, but it can potentially pose issues with smaller groups.
- Block randomization. Researchers can break participants into equally-numbered sub-groups, or blocks. This method may be more ideal for smaller-scale studies to prevent extreme discrepancies in allocation.
- Stratified randomization. In stratified randomization, researchers group participants with similar characteristics or attributes amongst their peers (into “strata”). They then select assign participants within those strata to either control or treatment groups using another randomization technique, usually simple randomization. Stratified randomization can accurately reflect population-wide results because it accounts for common influential factors in the greater public.
Here’s a sample stratified randomization design for studying outcomes of community-based medicine:

Covariate adaptive randomization. Covariate adaptive randomization groups participants based on covariates, which are independent variables that might have an impact on the outcome of a trial, but which are not intended as the primary target of study. This is the most complex randomization model, and for that reason has garnered some criticism because of difficulties in practice and challenges for using the data in later studies.

How to Learn More About Clinical Trials From Child Neurology Center Of NWF
Our Child Neurology Center of Northwest Florida exists to serve the community, to educate our neighbors about the important work that we do, and to deliver expert neurological care to our patients. Navigating the ins and outs of clinical trials can be complex. If you have more questions about trial randomization to eliminate bias, please don’t hesitate to contact us. We’re dedicated to keeping the residents of Northwest Florida up to date on the latest clinical research impacting the growing field of child neurology.